Immune Evasion And Provocation By Mycobacterium tuberculosis
No Comments on Immune Evasion And Provocation By Mycobacterium tuberculosis Mycobacterium tuberculosis
Immune Evasion And Provocation By Mycobacterium tuberculosis
Mycobacterium tuberculosis, the causative agent of tuberculosis, has infected humankind for millennia. M. tuberculosis has been prevalent to establish infection, persist inside the host even with a strong immune response, and has a higher potential to get transmitted to uninfected individuals. The recent data brings the diverse response of mankind toward M. tuberculosis infection.
Dynamic changes are occurring within the cellular and intracellular niches of M. tuberculosis during infection and its treatment. In this blog, we discuss the immune evasion and provocation by Mycobacterium tuberculosis while its course of infection and with a molecular understanding, effective vaccines, and adaptive immunity.
Introduction
Mycobacterium tuberculosis, a respiratory pathogen is estimated to infect about one-fourth of the global population and has been killing human lives more than any other microorganism has ever in history. While Mycobacterium tuberculosis originates from the Horn of Africa, before than seventy thousand years ago.
Mycobacterium tuberculosis has been ever evolving with millennia of changes in the immune system. The life cycle of M. tuberculosis persists on its higher ability to adapt and interact with the immune system in varied ways as follows:
- Evades Innate Immune Response
- Persists Face of Adaptive Immune Response being Asymptomatic
- Elicits a Robust Inflammatory Response causing Tissue Pathology
Understanding the pathology and molecular Mycobacterium tuberculosis, there is a crucial life cycle associated with it, considering the preventive and therapeutic vaccines, novel therapies, and biomarkers.
Strategies Of Immune Evasion and Provocation Of Mycobacterium tuberculosis
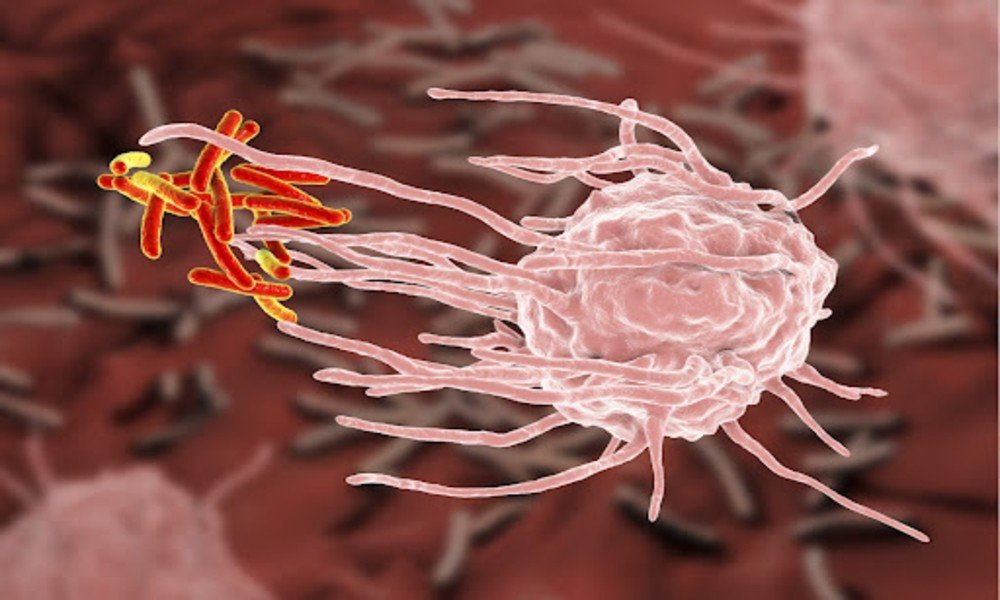
Decades of work at defining Mycobacterium tuberculosis resists and impairs the immune system’s myriad of macrophage defenses. With the impressive ability to impair phagolysosomal fusion, M. tuberculosis is now appreciated as a microorganism that survives diverse intracellular environments.
The early events of infection are crucial when preventive vaccines are under development. With the infectious dose being minimal as to three bacilli, these highlight how effective the Mycobacterium tuberculosis is while evading a human immune system. Also, how remarkably effective when it comes to disarming the innate immune system response resulting in successful evasion.
Molecular understanding of M. tuberculosis undermines the macrophage functions comes extensively from ex vivo studies, adding efforts in developing more relevant systems. Immune evasion strategies of Mycobacterium tuberculosis lays the groundwork for host-directed therapies the mycobacterial clearance.
Mycobacterium Tuberculosis Undermining Adaptive Immunity
- tuberculosis against having adaptive immune responses, even though robust, fails to sterilize the infection and delays the process of the immune response. In animal models, there is an antigen-specific T cell response detected in the lungs after several weeks of input. While in humans, there is a response detected after 2 to 8 weeks.
Conclusion
Tuberculosis has always been a disease that is a huge challenge, except it has been here for millennia. There has been vivid research and review, understanding the Mycobacterium tuberculosis multifaceted approach. The undermining approach of macrophage antimicrobial responses and an upper hand in potential survival in diverse intracellular environments.
We at GeNext Genomics serve the industry with high-quality biologics that help in excelling the country’s Biopharma Drug Discovery. Along with the production of biologics, there are proteins like STAT3, which satisfy every aspect of the research and development field.
With a talented team of professionals and expert scientists, GeNext Genomics takes pride in serving the industry by delivering great products.