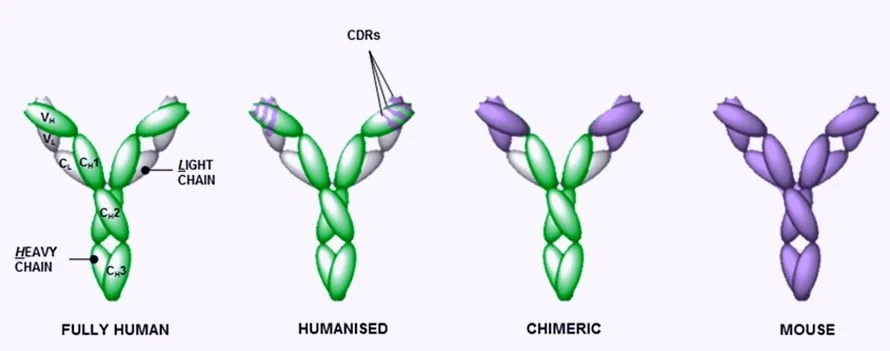
Chimeric And Humanized Antibodies are monoclonal antibodies that are non-human and are set into production by mice or rodents under specific laboratory conditions. Chimeric antibody usually tracks down and illuminize the tumour regions, whereas the humanized antibodies antigenic region is derived from human DNA and variable region from mouse or rodent DNA. These two have the standard function of administering to humans as anticancer drugs.
What is Chimeric Antibody?
A chimeric antibody is a non-human antibody variable region made by multiple species domains such as mouse, rat, Rabbit etc. It is fused with the Constant region of humans or any other species. For a quick example, the FC region of the mouse monoclonal antibody can be replaced by those regions of human antibody or another existing species of antibody. The chimeric antibody has the constant human domain and variable domain of the mouse that are cheaper than humanized antibodies.
A chimeric antibody can be made to retain the original antibody’s antigen specificity and affinity successfully. They are the most valuable tool for both in vivo and in vitro research and development for the diagnostic assay. These antibodies, if functionally active, can often be taken to therapeutic level.
These are examples of Chimeric antibodies in therapeutic areas: infliximab, rituximab, and abciximab. Infliximab, also known as Remicade, is used to treat rheumatoid arthritis, whereas rituximab, also known as Rituxan, is used to treat cancer.
What is Humanized Antibody?
A humanized antibody is a non-human antibody whose protein sequences are modified to enhance their similarity to humans’ naturally occurring antibody variants. Humanized antibodies play a crucial role by providing novel approaches to treating a broad range of diseases.
Humanized antibodies result from the process of humanization, which is a critical step in producing therapeutic antibodies. The process involves transferring or grafting vital non-human amino acids over a human antibody framework.
Fundamentally, the mouse amino acids in the CDRs or complementarity determining regions are transferred to the human variable framework regions. The resultant antibody created is known as a humanized antibody.
When under production, the human content is introduced more than the Chimeric antibody to reduce the risk associated with immunogenicity. There is also an introduction of the non-human to maintain the parent antibody’s primary binding activity. Humanized antibodies are originally made in other species and converted into human IgG. The technology reduces Human Anti-Mouse Antibodies (HAMA) respone, reducing immunogenicity and hence side effects.
Humanized monoclonal antibodies are used to treat various health conditions, even cancer. When compared to the Chimeric antibody, these humanized antibodies can reduce the immunogenic potential in the in vivo laboratory process. Trastuzumab or Herceptin is the first humanized antibody developed to treat breast cancer.
The Crucial Similarities Between Chimeric and Humanized Antibodies
- Chimeric and humanized antibodies are non-human antibodies.
- They are from non-human sources.
- Both have modified protein sequences which make them more similar to human antibodies.
- Monoclonal antibodies are produced primarily by mice and rats.
- These have Human DNA Derived antigenic region and a mouse/ rodent Derived variable region.
- They are therapeutic antibodies that humans can use.
- Moreover, they treat various conditions, including cancers, hence an anticancer drug.
- These antibodies can be used as controls for preclinical lead identification and potency assays to develop novel therapeutics.
- Both of them are highly target-specific.
The use of monoclonal antibodies as therapeutics has always had a long history. The Chimeric antibody and humanized antibody being developed for the same is impressive even when considering the scientific terms.
We at GeNext Genomics cater to all such monoclonal antibody needs, be it therapeutics like the Chimeric antibody or humanized antibody, for that matter. GNG has a human and mouse antibody library that can be used to cater to your need to make all formats of antibodies. We focus on delivering high-quality biologics put under production through our experts at work. We take care of the laboratory procedures with our keen scientists and their professional training peers.
Also Read: What Is Recombinant Protein Expression?